Chain Architecture, Crystallinity, and the Physical Basis of Polymer Performance
1 Introduction
The beauty of polymer chemistry lies not only in the reactions that join monomers but in the architecture that emerges afterwards.
Two polymers with the same chemical repeat unit can behave very differently if their chains are arranged, folded, or cross-linked in contrasting ways.
This lecture examines how molecular structure translates into mechanical, thermal, and optical properties. We will move from the level of covalent bonding to the supramolecular world of crystallites, amorphous regions, and phase separation, which together determine whether a polymer behaves like rubber, plastic, or fibre.
2 Levels of Structure in Polymers
Polymer scientists describe structure at several hierarchical levels:
| Level | Scale | Description | Key Techniques |
| Primary | Å nm | Chemical constitution: monomer type, configuration, tacticity | NMR, IR |
| Secondary | nm 10 nm | Chain conformation and local ordering (helices, folds) | X-ray diffraction, modelling |
| Tertiary | 10 100 nm | Packing of chains into crystallites, lamellae, or amorphous zones | SAXS, TEM |
| Quaternary / Morphological | > 100 nm | Domain organisation, spherulites, blends, composites | Optical microscopy, AFM |
Each level influences the next. Understanding these relationships enables predictive design of materials.
3 Molecular Weight and Chain Length
3.1 Degree of Polymerisation (DP)
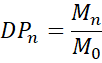
where Mn is the number-average molecular weight, and M0 is the molar mass of the repeat unit.
- Short chains (low DP) → lower strength, often wax-like.
- High DP (> 10⁴) → entanglement and high tensile strength.
3.2 Molecular-Weight Distribution (MWD)
The ratio
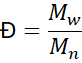
describes dispersity.
A narrow distribution (Đ ≈ 1.1–1.3) yields predictable melt behaviour; broader MWD provides processability but less uniformity.
3.3 Effect on Properties
- Tensile strength increases with molecular weight until entanglement saturation.
- Viscosity rises sharply with Mw3.4 (empirical relation)
- Impact resistance and film-forming ability depend on chain length continuity.
4 Chain Architecture
4.1 Linear Polymers
Simple chains without branches.
- High crystallinity is possible (e.g. HDPE).
- Pack efficiently; strong intermolecular forces.
4.2 Branched Polymers
Side chains disrupt packing:
- Lower density and melting point (e.g. LDPE).
- Greater flexibility and transparency.
4.3 Cross-linked (Network) Polymers
Chains joined by covalent bonds at junction points:
- Thermosets (epoxy, vulcanised rubber).
- Insoluble, infusible, dimensionally stable.
- Degree of cross-linking controls rigidity.
4.4 Star, Comb, and Dendritic Polymers
Special architectures created synthetically:
| Type | Description | Example | Behaviour |
| Star | Several linear arms from one core | Poly(styrene)-star-polyisoprene | Low viscosity melts |
| Comb / Brush | Long backbone with side chains | Poly(ethylene-co-vinyl acetate) | Soft, lubricious surfaces |
| Dendrimer | Repeated branched generations | PAMAM dendrimer | Precise size, functional end-groups |
Architecture, therefore, tunes flow, mechanical response, and surface chemistry.
5 Stereochemistry and Tacticity
5.1 Definition
Tacticity refers to the arrangement of substituents along a chain:
- Isotactic: all groups on the same side.
- Syndiotactic: alternating sides.
- Atactic: random orientation.
5.2 Consequences
| Polymer | Isotactic Form | Atactic Form |
| Polypropylene | Crystalline, tough | Amorphous, rubbery |
| Polystyrene | Crystallises slowly | Glassy and brittle |
| PMMA | Higher Tg when isotactic | Lower Tg and transparent |
Catalysts such as Ziegler–Natta or metallocenes enable stereoregular polymerisation and hence controlled morphology.
6 Conformation and Chain Flexibility
A polymer chain in solution or melt behaves like a random coil. Its statistical dimensions depend on:
- Bond rotation barriers (torsional potential).
- Intermolecular interactions (solvent quality).
- Temperature.
The radius of gyration Rg and the end-to-end distance ⟨R2⟩1/2 describe the size of a polymer coil. Stiffer chains (e.g., aramids) have a longer persistence length, which results in a high modulus but poor flexibility
7 Crystallinity in Polymers
7.1 What Is Crystallinity?
Regions where chains adopt ordered, repeating patterns are called crystallites.
They coexist with disordered amorphous regions.
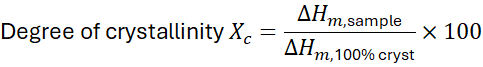
Measured via Differential Scanning Calorimetry (DSC).
7.2 Crystallisation Mechanism
- Nucleation: small ordered clusters form.
- Growth: lamellae extend outward, often radiating into spherulites.
- Perfection: rearrangement improves packing.
Crystallisation depends on cooling rate, molecular symmetry, and tacticity.
7.3 Examples
| Polymer | Crystallinity | Features |
| HDPE | High (70–90 %) | Opaque, high-strength |
| LDPE | Low (40 %) | Flexible, transparent |
| Nylon-6,6 | Moderate | Hydrogen bonding stabilises crystals |
| PET | Semi-crystalline | Controlled transparency for bottles |
8 Amorphous Regions
Chains in amorphous domains are randomly coiled.
Their thermal signature is a glass-transition temperature (Tg) where mobility changes abruptly.
- Below Tg: hard, brittle (glassy).
- Above Tg: soft, rubbery (viscoelastic).
Polystyrene → Tg ≈ 100 °C; PMMA ≈ 105 °C; Polyethene ≈ −125 °C.
Additives and plasticisers lower Tg by increasing free volume.
9 Morphological Structures
9.1 Spherulites
Under polarised light, semi-crystalline polymers display Maltese-cross patterns.
Each spherulite consists of radially growing lamellae separated by amorphous zones.
Size (1–100 µm) depends on nucleation density and cooling rate; smaller spherulites yield higher clarity and toughness.
9.2 Lamellae and Folds
Individual lamellae are ~10 nm thick; chains fold back and forth. This folded-chain model explains the balance between order and flexibility.
9.3 Orientation and Drawing
When polymer fibres are drawn, chains align along the stretch direction.
Consequences:
- Increased modulus and strength.
- Birefringence (optical anisotropy).
- Reduced elongation at break.
This principle underlies the production of Kevlar and PET fibres.
10 Intermolecular Forces
- Van der Waals / Dispersion: dominates in non-polar polymers (PE, PP).
- Dipole–dipole: important in PVC, PAN.
- Hydrogen bonding: in nylons, polyurethanes, PVA.
- π–π stacking: in conjugated systems (P3HT, polyaniline).
These forces dictate solubility, melting point, and mechanical coherence.
11 Thermal Transitions
| Transition | Description | Technique |
| Tg | onset of segmental mobility | DSC, DMA |
| Tm | melting of crystallites | DSC |
| Tc | crystallisation during cooling | DSC, hot-stage microscopy |
| Td | decomposition | TGA |
A typical semi-crystalline polymer exhibits Tg < T < Tm window where it is processable.
12 Mechanical Behaviour
12.1 Elastic vs Plastic Response
At small strain, polymer chains extend reversibly; beyond the yield point, they slide past each other (plastic deformation).
12.2 Stress–Strain Profiles
| Type | Characteristic Curve | Example |
| Brittle | Sharp break after elastic region | Polystyrene |
| Tough / Ductile | Yield, necking, draw, hardening | HDPE |
| Elastomeric | Large reversible strain | Natural rubber, silicone |
12.3 Time-Dependent Effects
Viscoelasticity arises because chain motion spans a range of relaxation times.
Measured by Dynamic Mechanical Analysis (DMA), providing storage (E′) and loss (E″) moduli.
13 Optical and Barrier Properties
- Transparency depends on refractive-index uniformity and crystal size < visible wavelength.
- Birefringence reveals orientation.
- Gas permeability decreases with higher crystallinity or polar interactions.
Thus, amorphous PET (bottle grade) is transparent, whereas highly crystalline PET (fibre grade) is opaque and strong.
14 Effect of Additives and Fillers
- Plasticisers: increase flexibility (PVC + phthalates).
- Fillers: improve stiffness or thermal stability (carbon black, talc).
- Reinforcing fibres: yield composites (glass fibre–epoxy).
- Nucleating agents: control spherulite size and transparency.
Morphology is engineered as much by formulation as by polymerisation.
15 Characterisation Techniques
| Method | Structural Information | Typical Outcome |
| Wide-Angle X-ray Scattering (WAXS) | Crystal lattice spacing | % crystallinity |
| Small-Angle X-ray Scattering (SAXS) | Lamellar spacing | Long-period structure |
| Differential Scanning Calorimetry (DSC) | Tg, Tm, Tc | Thermal map |
| Thermogravimetric Analysis (TGA) | Decomposition profile | Stability |
| Dynamic Mechanical Analysis (DMA) | Viscoelastic spectrum | E′, E″ vs T |
| Optical / Polarised Microscopy | Spherulites, orientation | Morphology |
| Transmission Electron Microscopy (TEM) | Nanostructure in block copolymers | Phase domains |
16 Case Studies
16.1 Polyethene
- Linear HDPE → high crystallinity → rigid containers.
- Branched LDPE → low crystallinity → film applications.
- Copolymerisation with α-olefins (LLDPE) tunes flexibility.
16.2 Polyethene Terephthalate (PET)
- Drawn, oriented PET fibres → high strength.
- Semi-crystalline PET bottles → dimensional stability and clarity.
16.3 Nylon-6,6
Hydrogen-bonded sheets form robust crystals; heat-setting improves moisture resistance and mechanical retention.
16.4 Elastomers (Natural Rubber, SBR)
Amorphous chains with limited cross-linking retract rapidly after deformation.
Entropy elasticity rather than bond stretching governs behaviour.
17 Structure–Property–Processing Triangle
A guiding principle in materials science:
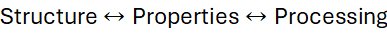
- Cooling rate → crystal size.
- Orientation → strength.
- Annealing → defect healing.
Designers must therefore control molecular and processing parameters concurrently.
18 Emerging Morphological Concepts
- Nanocomposites: exfoliated clay or graphene sheets within polymer matrices enhance modulus and barrier properties.
- Self-healing networks: reversible bonds (Diels–Alder, hydrogen bonding).
- Shape-memory polymers: dual-phase morphology (hard and soft domains).
- Liquid-crystalline polymers: ordered mesophases between amorphous and crystalline states.
- Conducting polymers: π-stacked domains allow charge transport while retaining flexibility.
19 Environmental and Recycling Considerations
Morphology affects recyclability:
- Highly cross-linked thermosets resist remelting.
- Semi-crystalline thermoplastics can be remoulded if degradation is limited.
- Research into vitrimers introduces dynamic covalent links enabling re-processability.
Control of crystallinity also influences biodegradation rates in polyesters and polyhydroxyalkanoates (PHAs).
20 Summary
| Structural Parameter | Controls | Typical Measurement |
| Molecular weight | Strength, viscosity | GPC |
| Branching | Density, flexibility | NMR |
| Tacticity | Crystallinity | NMR, XRD |
| Crystallinity | Modulus, barrier | DSC, WAXS |
| Orientation | Tensile strength | X-ray, birefringence |
| Cross-link density | Elastic modulus | Swelling tests |
Polymer morphology is thus the bridge between chemical composition and engineering application.
Mastery of this bridge allows chemists to sculpt materials with precision, from transparent films to bulletproof fibres.
21 Further Reading and Live Learning Links
- Royal Society of Chemistry: Polymer Morphology Overview
- Polymer Science Learning Centre: Crystallinity and Amorphous Regions
- Chemguide: Polymer Structures and Uses
- Khan Academy: Structure and Properties of Polymers
- Science History Institute: Nylon and Polymer Morphology
These provide accessible visuals and experimental demonstrations, complementing this lecture.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections


