Designing Random, Block, Alternating, and Graft Systems for Tunable Properties
1 Introduction
Most polymers studied so far consist of a single repeating unit, derived from one monomer.
While such homopolymers (e.g. polyethene, polystyrene) have well-defined traits, they can be limited in versatility. To tailor elasticity, hardness, permeability, or chemical resistance, chemists combine two or more monomers within the same chain.
This strategy is called copolymerisation. It allows the deliberate design of polymers whose average composition and microstructure sit between those of their constituent homopolymers or, in some cases, exhibit entirely new behaviours.
Copolymers are ubiquitous: from ABS plastics (acrylonitrile–butadiene–styrene) to styrene–butadiene rubber and ethylene–vinyl acetate. Understanding how monomers interact during polymerisation, and how their arrangement governs properties, is key to both synthetic design and industrial formulation.
2 Basic Concepts
2.1 Definition
A copolymer is a polymer derived from two or more different monomer species.
When A and B represent two distinct monomers:
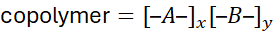
where x and y may vary randomly, alternately, or in blocks.
2.2 Terminology
| Term | Meaning |
| Binary copolymer | Formed from two monomers (most common). |
| Ternary / quaternary copolymer | Formed from three or more monomers. |
| Composition ratio | Molar fraction of each monomer in the chain. |
| Sequence distribution | The order in which A and B units occur. |
| Architectural design | Whether chains are linear, branched, grafted, or networked. |
3 Types of Copolymers
3.1 Random (Statistical) Copolymers
Monomers are incorporated at random along the chain, depending on their reactivity ratios.
Example: Styrene–acrylonitrile (SAN).
- Structure: –A–A–B–A–B–B–A–A–
- Formation: Simultaneous polymerisation of both monomers with similar radical reactivity.
- Properties: Intermediate between A and B; typically amorphous, transparent, and tough.
3.2 Alternating Copolymers
Monomers alternate in a regular ABAB pattern, often when each prefers to react with the other:
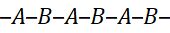
- Typical example: Maleic anhydride–styrene copolymer.
- Mechanism: Strong donor–acceptor interaction between monomers (electron-rich and electron-poor).
- Result: Uniform composition; well-defined glass-transition temperature (Tg).
3.3 Block Copolymers
Chains composed of long sequences (blocks) of one monomer followed by another:
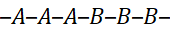
- Synthesis: Sequential living polymerisation (e.g. anionic, RAFT).
- Behaviour: Microphase separation into nanodomains (A-rich and B-rich).
- Applications: Thermoplastic elastomers, surfactants, and nanostructured films.
- Example: Polystyrene-block-polybutadiene-block-polystyrene (SBS).
3.4 Graft Copolymers
A backbone polymer of one type carries side chains (branches) of another:
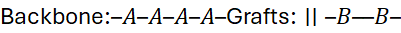
- Preparation: “Grafting-onto”, “grafting-from”, or “grafting-through” methods.
- Utility: Combines the flexibility of one component with the strength or compatibility of another.
- Example: Natural rubber grafted with polystyrene (HIPS).
3.5 Gradient and Statistical Block Copolymers
Modern controlled polymerisation allows gradual composition change along the chain, useful for compatibilisers in blends and emulsions.
4 Reactivity Ratios and Copolymer Composition
4.1 The Reactivity-Ratio Model
For radical copolymerisation of monomers M₁ and M₂,
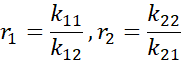
The symbol kᵢⱼ represents the rate constant for a polymer chain that ends with monomer i to add a monomer j.
- If both r₁ and r₂ equal 1, the copolymer formed is a random copolymer.
- If the product of r₁ and r₂ equals 1, the system is azeotropic, meaning the copolymer composition remains constant during polymerisation.
- If both r₁ and r₂ are approximately 0, the copolymer tends to be alternating.
- If both r₁ and r₂ are much greater than 1, the polymer has blocky or homopolymer-rich sequences.
4.2 The Mayo–Lewis Equation
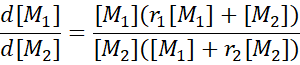
This relation predicts instantaneous composition and enables graphical determination of r₁ and r₂ from experimental data (Fineman–Ross or Kelen–Tüdös methods).
5 Factors Affecting Copolymer Structure
- Monomer polarity and resonance donor–acceptor interactions favour alternation.
- Steric effects of bulky substituents hinder successive identical additions.
- Solvent and temperature influence radical stability and diffusion.
- Initiator system ionic vs radical can change the preference for one monomer.
- Feed ratio composition drift occurs as one monomer is consumed faster.
6 Synthesis Methods
6.1 Bulk and Solution Copolymerisation
- Simple but heat-management critical.
- Useful for random and alternating copolymers.
6.2 Emulsion and Suspension Polymerisation
- Allow control of particle size and heat removal.
- Industrially used for ABS, SBR, and vinyl acetate copolymers.
6.3 Controlled/Living Techniques
Living radical systems (ATRP, RAFT, NMP) and anionic routes enable:
- Sequential addition for block copolymers.
- Post-functionalisation via chain ends.
- Narrow molecular-weight distribution.
6.4 Grafting Techniques
| Method | Description | Example |
| Grafting-onto | Pre-formed chains are attached to the backbone via reactive sites. | PEG onto PLA for hydrophilicity. |
| Grafting-from | Side-chain growth initiated from backbone sites. | PS grafts from cellulose. |
| Grafting-through | Copolymerisation of macromonomers bearing polymerisable groups. | PMMA-g-PDMS. |
7 Examples of Industrial Copolymers
| Copolymer | Composition | Type | Key Properties | Applications |
| SBR | Styrene + Butadiene | Random | Tough, flexible, abrasion-resistant | Tyres, footwear |
| ABS | Acrylonitrile + Butadiene + Styrene | Graft | Rigid, glossy, impact-resistant | Appliance housings |
| EVA | Ethylene + Vinyl acetate | Random | Soft, transparent, flexible | Films, adhesives |
| NBR | Acrylonitrile + Butadiene | Random | Oil-resistant | Seals, hoses |
| SMA | Styrene + Maleic anhydride | Alternating | Reactive sites for modification | Engineering plastics |
| PE-co-PP | Ethylene + Propylene | Statistical | Elastic, weather-resistant | Automotive parts |
8 Structure–Property Relationships
8.1 Thermal Behaviour
- Random copolymers often have a single glass-transition temperature (Tg) between those of the homopolymers.
- Block copolymers may exhibit two Tgs, corresponding to microphase domains.
- Alternating copolymers can crystallise if sequence regularity allows.
8.2 Mechanical Properties
- Hardness and flexibility can be tuned by adjusting block length or composition.
- SBS and SEBS block copolymers act as thermoplastic elastomers, solid at room temperature, and processable at high temperatures.
8.3 Optical and Barrier Properties
- Incorporating polar monomers (acrylonitrile, vinyl acetate) increases transparency and gas-barrier strength.
- Grafting hydrophilic chains (PEG) onto hydrophobic backbones yields amphiphilic polymers useful in coatings and biomedical devices.
9 Microphase Separation in Block Copolymers
9.1 Concept
In block copolymers, chemically distinct blocks are thermodynamically immiscible but covalently joined. The resulting compromise produces ordered nanostructures, spheres, cylinders, and lamellae depending on composition and interaction parameter (χ).
9.2 Theoretical Framework
The Flory–Huggins interaction parameter (χ) and volume fraction (f) of each block determine morphology:
- fₐ ≈ 0.5 → lamellae
- fₐ < 0.3 → cylinders
- fₐ < 0.1 → spheres
9.3 Applications
- Nanolithography templates (PS-b-PMMA)
- Drug-delivery micelles (PEG-b-PLA)
- Ion-conducting membranes (PEO-b-PS sulfonated systems)
10 Compatibilisation of Polymer Blends
Copolymers often serve as interfacial compatibilisers between immiscible polymers.
For example, adding a styrene–maleic anhydride copolymer improves adhesion between PS and polyamide.
Mechanism:
- One segment dissolves in each phase, reducing interfacial tension.
- Improved morphology leads to enhanced mechanical strength.
11 Analytical and Characterisation Techniques
| Technique | Purpose | Information Gained |
| NMR (¹H, ¹³C) | Sequence distribution, block length | Ratio of A:B units |
| FT-IR | Functional-group identification | Presence of comonomer units |
| DSC / DMA | Thermal transitions | Tg values, phase separation |
| GPC (SEC) | Molecular-weight distribution | Dispersity (Ð) |
| SAXS / TEM | Morphology of block copolymers | Domain spacing |
| Elemental / composition analysis | Average copolymer composition | Feed vs actual incorporation |
12 Sustainability and Bio-based Copolymers
Modern polymer science increasingly integrates renewable monomers and degradability:
- PLA-co-PEG: enhances the flexibility and hydrophilicity of polylactide.
- PBS-co-PBAT: combines biodegradability with ductility.
- Bio-PE-co-Bio-PP: from bio-ethanol feedstocks.
- CO₂-epoxide copolymers: produce polycarbonates as CO₂ sinks.
These materials bridge performance with environmental responsibility.
13 Mathematical Modelling of Copolymerisation
Advanced simulation approaches (Monte Carlo, kinetic models) predict sequence distribution and molecular-weight evolution under varying conditions.
Key outputs:
- Instantaneous copolymer composition
- Drift in feed ratio
- Composition profile along chain length
Such modelling aids process control in industry, ensuring reproducibility and targeted properties.
14 Recent Research Trends
- Sequence-controlled polymers using programmable monomer addition.
- Click-chemistry post-functionalisation for modular design.
- Reversible deactivation radical polymerisation (RDRP) for multi-block systems.
- Stimuli-responsive copolymers (temperature, pH, light).
- Recyclable and dynamic covalent networks derived from block copolymers.
15 Summary
| Copolymer Type | Synthesis Route | Morphology | Key Properties | Typical Uses |
| Random | Simultaneous radical polymerisation | Disordered | Intermediate flexibility | General plastics |
| Alternating | Electron donor–acceptor pairs | Regular AB sequence | Defined Tg, uniformity | Engineering resins |
| Block | Sequential living polymerisation | Microphase-separated | Elastomeric, nanostructured | TPEs, membranes |
| Graft | Backbone + side-chain growth | Branched | Combined attributes | Impact modifiers |
| Gradient | Controlled feed variation | Smooth composition | Compatibility tuning | Blend compatibilisers |
Copolymers thus offer a toolbox for designing materials that span multiple property domains without synthesising new monomers from scratch.
16 Further Reading and Live Learning Links
- Polymer Science Learning Centre: Copolymers
- Royal Society of Chemistry: Copolymers Explained
- Chemguide: Addition Polymerisation and Copolymers
- Science History Institute: Synthetic Rubber Story (SBR)
- Khan Academy: Polymers and Copolymers
Each resource provides accessible diagrams, historical insights, and industrial relevance for further study.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections