A companionable guide to chemical equilibrium and system response with modular clarity and outreach-ready tone
Why equilibrium matters
Chemical reactions don’t always go to completion; many reach a dynamic equilibrium, where forward and reverse reactions occur at equal rates. Le Chatelier’s Principle helps us predict how a system at equilibrium responds to changes in:
- Concentration
- Pressure
- Temperature
- Volume
Understanding these shifts is essential for:
- Industrial optimisation (e.g. ammonia synthesis)
- Laboratory control
- Environmental and biological systems
Explore foundational concepts at Chemguide – Le Chatelier’s Principle.
What is Le Chatelier’s Principle?
Le Chatelier’s Principle states:
“If a system at equilibrium is disturbed, the system will adjust to counteract the disturbance and restore equilibrium.”
This principle helps predict the direction of shift, either favouring the forward reaction (more products) or the reverse reaction (more reactants).
1. Change in Concentration
- Adding reactants → shifts the equilibrium right (forms more products)
- Removing reactants → shifts the equilibrium left (forms more reactants)
- Adding products → shifts the equilibrium left
- Removing products → shifts the equilibrium right
Example:
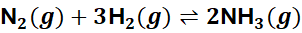
Adding H₂ shifts the equilibrium right → more NH₃ formed.
2. Change in Pressure (gaseous systems only)
- Increasing pressure → shifts the equilibrium toward the side with fewer gas molecules
- Decreasing pressure → shifts the equilibrium toward the side with more gas molecules
Example:
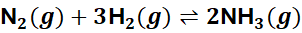
There are 4 moles of gas on the left and 2 moles on the right.
→ Increasing pressure shifts the equilibrium right, favouring NH₃ formation.
3. Change in Temperature
- Increasing temperature → favours the endothermic direction
- Decreasing temperature → favours the exothermic direction
Example:
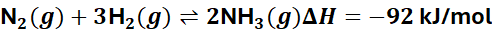
This reaction is exothermic (releases heat).
→ Increasing temperature shifts equilibrium left → less NH₃ formed.
4. Change in volume (gaseous systems)
- Decreasing volume → increases pressure → same effect as pressure increase
- Increasing volume → decreases pressure → same effect as pressure decrease
Visualising equilibrium shifts
Use concentration-time graphs to show:
- Initial disturbance
- Direction of shift
- New equilibrium position
These graphs help learners see dynamic adjustment and stabilisation.
Explore visual tools at Khan Academy – Equilibrium Shifts.
Industrial applications
Le Chatelier’s Principle guides optimisation in:
- Haber process (ammonia synthesis)
- Contact process (sulfuric acid production)
- Methanol synthesis
Conditions are chosen to maximise yield while balancing cost and rate:
- High pressure favours product formation
- Moderate temperature balances yield and reaction rate
- Catalysts speed up equilibrium attainment without shifting position
Explore industrial case studies at the Royal Society of Chemistry – Equilibrium in Industry.
Common misconceptions
- Equilibrium doesn’t mean equal amounts; it means equal rates
- Catalysts don’t shift the equilibrium; they only speed up attainment
- Le Chatelier’s Principle applies only to systems at equilibrium
- Pressure changes only affect gaseous reactions
- Temperature changes affect both rate and position
Clarifying these points supports deeper understanding and practical application.
Closing: Equilibrium as dialogue
Le Chatelier’s Principle reveals the responsive nature of chemical systems, always adjusting, always seeking balance. By understanding these shifts, we can predict, control, and optimise reactions in both lab and life.
This lecture equips you to:
- Apply Le Chatelier’s Principle to various disturbances
- Predict equilibrium shifts in concentration, pressure, temperature, and volume
- Interpret graphs and industrial strategies
- Clarify common misconceptions
- Connect equilibrium dynamics to real-world systems
Available in PDF format with an associated quiz
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections