Mechanisms, Kinetic Principles, and Material Applications in Chain-Growth Systems
1. Introduction
Having examined step-growth polymerisation in Lecture 3, we now turn to a complementary mechanism, chain-growth polymerisation, focusing on its most widespread variant: free radical polymerisation.
This reaction type underpins the production of the world’s most common plastics, such as polyethene, polystyrene, poly(vinyl chloride), and poly(methyl methacrylate). Its versatility, tolerance to functional groups, and ability to proceed under relatively mild conditions make it central to both industrial synthesis and academic study.
In this lecture, we will explore:
- The chemical nature of radicals and how they initiate polymer growth.
- The three fundamental stages are initiation, propagation, and termination.
- The kinetic and thermodynamic principles governing molecular weight and rate.
- Methods for controlling radical reactions, including controlled/living polymerisation.
- The applications, limitations, and sustainability aspects of free radical polymers.
2. Conceptual Overview of Chain-Growth Polymerisation
2.1 What Defines Chain-Growth?
In chain-growth polymerisation:
- Initiation generates an active species (radical, cation, or anion).
- Propagation adds monomer units rapidly to the growing chain.
- Termination ends growth, either by combination or disproportionation.
Only monomers react with active centres; the growing chain does not react with other chains or oligomers, unlike in step-growth systems. This gives rise to rapid molecular weight build-up early in the reaction, even at low conversion.
2.2 Typical Monomers
Free radical polymerisation requires unsaturated monomers containing a carbon–carbon double bond:
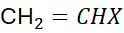
where X is an electron-withdrawing or neutral substituent such as Cl, CN, phenyl, or CO₂CH₃.
Examples:
| Monomer | Structure | Polymer |
| Ethene | CH₂=CH₂ | Polyethene |
| Vinyl chloride | CH₂=CHCl | PVC |
| Styrene | CH₂=CHPh | Polystyrene |
| Methyl methacrylate | CH₂=C(CH₃)CO₂CH₃ | PMMA |
3. Radical Formation and Reactivity
3.1 What is a Free Radical?
A free radical is a species with an unpaired electron, typically generated by homolytic bond cleavage.
Radicals are highly reactive, short-lived intermediates that initiate and propagate polymer chains through addition to C=C bonds.
3.2 Radical Sources
Radicals can be produced by:
- Thermal decomposition of peroxides or azo compounds
- Benzoyl peroxide → 2 PhCO·O → 2 Ph· + 2 CO₂
- AIBN (azobisisobutyronitrile) → 2 R· + N₂
- Photochemical initiation using UV light
- Useful in coatings and photopolymerisation.
- Redox initiation
- Common in aqueous systems, e.g. persulfate with an iron(II) salt.
Each initiation route is chosen according to temperature range, solvent compatibility, and required reaction rate.
4. The Mechanism of Free Radical Polymerisation
The full reaction pathway consists of three key stages.
4.1 Initiation
Two sub-steps:
Step 1: Initiator decomposition (Initiation)
The initiator, often a diatomic molecule like I – I (iodine), decomposes to form radicals:
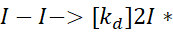
- Kd is the decomposition rate constant of the initiator.
- I* represents an iodine radical.
- The reaction produces two radicals from one initiator molecule.
This is often written as:
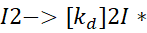
This step is crucial because radicals are highly reactive species that can start the polymerisation.
Step 2: Radical addition to monomer (First propagation step)
The radical generated from the initiator reacts with a monomer, e.g., CH2 = CHX:
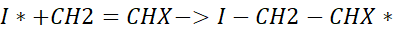
- The iodine radical attacks the double bond of the monomer.
- This creates a new radical at the end of the monomer unit, which is now the first active chain radical.
This new radical can continue to react with more monomer molecules in the propagation steps, growing the polymer chain.
Summary in words:
- Initiation: The initiator decomposes to form radicals.
- First propagation: A radical adds to the monomer, forming the first active chain radical that will propagate the polymer chain.
This produces the first active chain radical.
The rate of initiation:
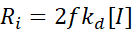
where f is the efficiency (fraction of radicals actually initiating chains).
4.2 Propagation
The growing radical reacts with successive monomer units:
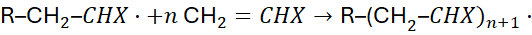
Each propagation step has the same rate constant, kₚ.
This phase continues rapidly, adding thousands of monomers per second.
4.3 Termination
Growth ceases when two radical species react:
- Combination:
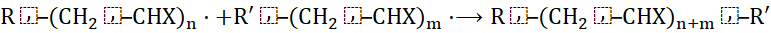
Disproportionation:
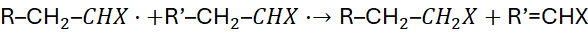
Both remove radicals from the system.
The overall termination rate:
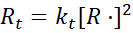
5. Kinetic Expressions and Steady-State Approximation
At steady state:
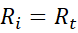
So, the concentration of radicals remains constant.
Combining rate laws gives:
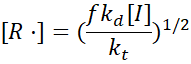
and the rate of polymerisation becomes:
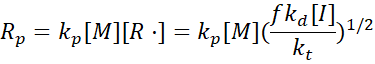
Hence, polymerisation rate depends on the square root of initiator concentration, a hallmark of radical kinetics.
6. Chain Length and Molecular Weight Control
6.1 Degree of Polymerisation (DPₙ)
Average chain length is determined by the ratio of propagation to termination events:
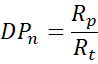
Since both depend on radical concentration, experimental control requires adjustment of temperature, initiator level, or chain-transfer agents.
6.2 Chain Transfer
Chain transfer reactions reduce molecular weight by generating new radicals mid-chain:
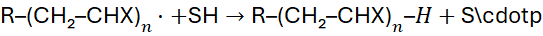
Common agents: mercaptans, solvents, or monomers themselves.
This technique allows molecular weight control and is crucial in making products such as low-density polyethene.
7. Thermodynamics of Radical Polymerisation
Despite being exothermic overall (ΔH ≈ –80 kJ mol⁻¹ per monomer unit), polymerisation reduces entropy as monomers become ordered in chains. Thus, the reaction becomes unfavourable at high temperature, defining a ceiling temperature (Tc) where ΔG = 0.
For styrene, Tc ≈ 310 °C; above this, depolymerisation dominates.
8. Structural Consequences of Radical Mechanism
Radical polymerisation tends to yield atactic polymers with a random stereochemical configuration at the chiral centres because radical addition is not stereospecific.
| Polymer | Typical tacticity | Resulting property |
| Polystyrene | Atactic | Amorphous, brittle |
| Polypropylene (radical) | Atactic | Non-crystalline |
| PMMA | Atactic | Transparent |
To obtain isotactic or syndiotactic order, ionic or coordination polymerisation methods are required (see Lecture 5).
9. Industrial Examples
9.1 Polyethene (PE)
Two major radical-based processes:
- Low-density polyethene (LDPE):
Formed at 1000–3000 bar and 200–300 °C using oxygen or peroxide initiators.
The result is a highly branched polymer, soft and flexible. - High-pressure tubular or autoclave reactors:
Continuous systems that manage radical reactions safely under extreme conditions.
Applications: films, bags, insulation.
9.2 Polystyrene (PS)
Produced via thermal decomposition of benzoyl peroxide in bulk or suspension.
- Amorphous, transparent, rigid.
- Easily foamed (as expanded polystyrene).
- Susceptible to cracking solvents (e.g. acetone).
Applications: packaging, electronics casings, laboratory ware.
9.3 Poly(vinyl chloride) (PVC)
Polymerisation of vinyl chloride monomer (VCM) under suspension or emulsion conditions using peroxides or azo initiators.
- Contains chlorine substituents, enhancing flame resistance.
- Thermoplastic yet rigid unless plasticised.
Applications: pipes, flooring, window frames, and electrical cable sheaths.
9.4 Poly(methyl methacrylate) (PMMA)
Formed via radical polymerisation of methyl methacrylate using benzoyl peroxide.
- Transparent and UV-resistant (“acrylic glass”).
- Used for lenses, glazing, and signage.
10. Techniques and Reactor Types
| Method | Description | Typical use |
| Bulk polymerisation | Pure monomer + initiator | High purity, but heat removal is difficult |
| Solution polymerisation | Monomer dissolved in solvent | Easier temperature control |
| Suspension polymerisation | Droplets of monomer dispersed in water | Bead polymers (PVC, PS) |
| Emulsion polymerisation | Micelles of monomer stabilised by surfactant | Latex, coatings, adhesives |
Emulsion polymerisation is particularly efficient, producing high molecular weight polymers with fast kinetics and controllable particle size.
11. Copolymerisation under Radical Conditions
Radical mechanisms readily accommodate mixed monomer feeds, leading to random or alternating copolymers.
The composition follows the Mayo–Lewis equation involving reactivity ratios (r₁, r₂).
For example, styrene–butadiene copolymer (SBR) combines the rigidity of styrene with the elasticity of butadiene.
12. Controlled and Living Radical Polymerisation
Conventional radical polymerisation produces broad molecular weight distributions. Modern research has introduced controlled/living variants that suppress termination and transfer, enabling precision polymer design.
12.1 Atom Transfer Radical Polymerisation (ATRP)
- Catalytic system: Cu(I)/ligand complex mediates reversible halogen transfer.
- Allows predictable molecular weights and block copolymer formation.
12.2 Reversible Addition–Fragmentation Chain Transfer (RAFT)
- Utilises thiocarbonylthio compounds as mediators.
- Compatible with many monomers and solvents.
12.3 Nitroxide-Mediated Polymerisation (NMP)
- Stable nitroxide radicals reversibly cap the growing chain.
- Suitable for styrenic systems.
Controlled radical techniques bridge the gap between traditional radical and ionic/coordination methods, producing designer polymers for biomedical and nanotechnological applications.
13. Characterisation of Radical Polymers
| Technique | Purpose | Typical Observation |
| Infrared (IR) | Monomer disappearance (C=C stretch ~1640 cm⁻¹) | Confirms conversion |
| NMR spectroscopy | Chain structure and tacticity | Ratio of isotactic/atactic units |
| GPC (SEC) | Molecular weight distribution | Mₙ, Mₙ₊₁, PDI |
| DSC / TGA | Thermal transitions and stability | Tg, Tm, decomposition |
| Electron Spin Resonance (ESR) | Radical detection (transient) | Mechanistic study |
14. Environmental and Safety Considerations
14.1 Residual Monomers and Initiators
Incomplete conversion may leave unreacted monomers; purification or post-polymerisation treatments are required to reduce toxicity (e.g. residual styrene in polystyrene).
14.2 Thermal Management
Radical polymerisations are highly exothermic; inadequate cooling can cause runaway reactions. Industrial reactors are fitted with advanced temperature control and pressure relief systems.
14.3 Recycling and Degradation
Radical-based polymers (PE, PS, PVC) are generally non-biodegradable. Recycling strategies focus on:
- Mechanical recycling (melting and remoulding).
- Chemical recycling to monomers via pyrolysis.
- Designing degradable copolymers incorporating hydrolysable linkages.
15. Comparison: Radical vs Step-Growth Polymerisation
| Feature | Radical (Chain-Growth) | Step-Growth |
| Monomers | Vinyl (C=C) | Bifunctional (e.g. diacids, diols) |
| Growth mechanism | Active centre adds monomer | Stepwise condensation |
| By-products | None | Often small molecules |
| Rate of molecular weight increase | Early | Late |
| Molecular weight control | Via initiator and transfer | Via stoichiometry |
| Typical polymers | PE, PS, PVC, PMMA | Polyesters, nylons |
16. Applications and Modern Developments
16.1 Bulk Commodity Materials
- Packaging films and containers (PE)
- Building materials (PVC)
- Insulation and foams (PS, PMMA)
16.2 Coatings and Adhesives
Radical-cured systems dominate UV-curable coatings, dental resins, and pressure-sensitive adhesives.
16.3 Biomedical and Advanced Materials
Controlled radical polymerisation enables biocompatible hydrogels, drug-delivery systems, and surface grafting for implants.
16.4 Nanocomposites
Incorporating nanoparticles during radical polymerisation produces hybrid materials with improved barrier and mechanical properties.
17. Experimental Demonstration: Polystyrene Bead Formation
Suspension polymerisation experiment:
- Disperse styrene monomer and benzoyl peroxide in water with poly(vinyl alcohol) stabiliser.
- Heat to 80–90 °C with stirring.
- Observe spherical beads forming as polymerisation proceeds.
- Filter, wash, and dry to obtain solid polystyrene spheres.
This classic demonstration illustrates exothermic behaviour, initiation efficiency, and practical reactor control.
18. Advantages and Limitations
Advantages
- Simple, robust, and adaptable process.
- Broad monomer scope and tolerance to impurities.
- High polymerisation rate.
- Amenable to mass production.
Limitations
- Limited control of molecular weight distribution.
- Random stereochemistry (atactic).
- Thermal management challenges in bulk reactions.
- Environmental persistence of resultant polymers.
19. Summary and Key Takeaways
- Free radical polymerisation is the dominant chain-growth process, responsible for many everyday plastics.
- It proceeds through initiation, propagation, and termination steps involving reactive radicals.
- Kinetics show a square-root dependence on initiator concentration.
- Molecular weight is governed by termination and chain-transfer events.
- Modern controlled radical methods (ATRP, RAFT, NMP) permit near-living behaviour.
- Industrial processes rely on safe heat management and environmental consideration.
This mechanistic foundation prepares us for Lecture 5, which explores the ionic and coordination variants of chain-growth polymerisation, offering stereochemical precision beyond the radical process.
20. Further Reading and Live Learning Links
- Royal Society of Chemistry: Free Radical Polymerisation Overview
- Polymer Science Learning Centre: Radical Polymerisation
- Khan Academy: Chain-Growth Mechanisms
- Science History Institute: The Story of Plastics
- Chemguide: Addition Polymerisation
These links offer reaction animations, historical background, and detailed mechanistic discussions to support deeper engagement.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections


