Condensation Pathways, Mechanistic Insight, and Material Applications in Modern Polymer Chemistry
1. Introduction
In the previous lecture, we explored the structural and categorical diversity of polymers from their origins to their thermal behaviour. We now turn to a specific mechanism of polymer formation: step-growth polymerisation, historically known as condensation polymerisation.
Step-growth polymerisation is a cornerstone of synthetic polymer chemistry. It underlies the creation of some of the most widely used materials, polyesters, polyamides (nylons), and polyurethanes, which together shape industries as diverse as textiles, packaging, automotive engineering, and biomedical devices.
This lecture will examine the mechanistic logic, kinetic principles, molecular design, and industrial significance of step-growth polymerisation, highlighting both the elegance of its chemistry and the practicalities of its application.
2. Defining Step-Growth Polymerisation
2.1 Conceptual Overview
In step-growth polymerisation, any two reactive species, monomers, dimers, trimers, or longer oligomers can combine at any stage of the reaction. Unlike chain-growth polymerisation, which proceeds via active centres and adds monomers sequentially to a growing chain, step-growth involves gradual molecular build-up through bifunctional or polyfunctional monomers reacting stepwise.
A simple model is:
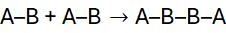
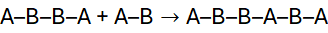
Each step forms longer chains until a high molecular weight polymer results, provided the reaction proceeds almost to completion.
2.2 Condensation Reactions
Step-growth polymerisation is frequently associated with condensation reactions, in which small molecules such as water, methanol, hydrogen chloride, or ammonia are released as by-products.
For example:
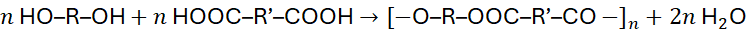
This represents the synthesis of a polyester from a diol and a dicarboxylic acid.
However, not all step-growth reactions involve elimination. Polyurethanes, for instance, form through the reaction of diols with diisocyanates, producing no by-product, a non-condensation step-growth process.
3. Historical Foundations
The theoretical understanding of step-growth polymerisation owes much to Wallace H. Carothers, who, in the 1930s, established the quantitative framework of polymer formation at DuPont. His pioneering work led to the invention of nylon, neoprene, and polyesters, and introduced the now-classic equation linking extent of reaction and degree of polymerisation.
Carothers’ insights demonstrated that high molecular weight can only be achieved when the reaction approaches near-complete conversion of functional groups, typically above 99%. This was a fundamental discovery, distinguishing step-growth from the rapid chain-length build-up observed in chain-growth processes.
4. Kinetic Features of Step-Growth Polymerisation
4.1 Reaction Sequence
Each condensation event in step-growth polymerisation is chemically identical and statistically possible between any reactive molecules. Therefore, there is no concept of “active centres” or propagation in the chain-growth sense.
The reaction rate depends on monomer concentration and the reactivity of functional groups. The general kinetic scheme can be summarised as:
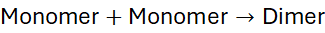
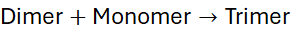
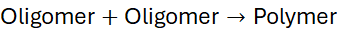
This statistical pattern gives rise to a broad molecular weight distribution and a gradual increase in viscosity as the reaction progresses.
4.2 Extent of Reaction (p)
The extent of reaction, p, is defined as the fraction of functional groups that have reacted. As pincreases, the average chain length increases sharply.

4.3 Carothers’ Equation
Carothers derived a simple yet powerful relationship between and the degree of polymerisation (DP):
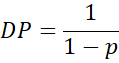
At 95% conversion (p = 0.95), (DP = 20), at 99%, DP = 100: and at 99.9%, DP – 1000.
Hence, almost complete reaction is required to achieve the high molecular weights characteristic of usable polymers.
Hence, almost complete reaction is required to achieve the high molecular weights characteristic of usable polymers.
5. Mechanistic Pathways and Functional Group Chemistry
5.1 Hydroxyl–Carboxyl Reactions (Polyesters)
Polyesters form via condensation between diols and dicarboxylic acids or their derivatives (acid chlorides, anhydrides).
Example:
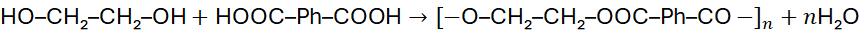
Poly(ethylene terephthalate), PET
Catalysts such as antimony trioxide or titanium(IV) butoxide enhance reaction rates.
5.2 Amine–Carboxyl Reactions (Polyamides / Nylons)
Polyamides, such as nylons, are produced through condensation between diamines and dicarboxylic acids.
Example:
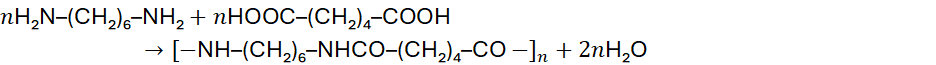
Nylon-6,6
Strong hydrogen bonding between amide linkages provides excellent strength and thermal stability.
5.3 Isocyanate–Hydroxyl Reactions (Polyurethanes)
Polyurethanes are formed by reaction between diisocyanates and diols:
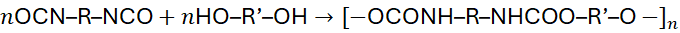
No by-product is released, but the polymer is still formed via a stepwise addition of functional groups.
5.4 Amine–Formaldehyde Condensations
Urea–formaldehyde and melamine–formaldehyde resins arise from condensation between amine groups and formaldehyde, producing thermosetting network structures used in coatings and laminates.
6. Molecular Design Considerations
6.1 Functionality
Functionality refers to the number of reactive sites per monomer molecule.
- Difunctional monomers (functionality = 2) produce linear polymers.
- Tri- or polyfunctional monomers lead to cross-linked or network polymers.
For example, glycerol (functionality = 3) reacting with phthalic anhydride produces alkyd resins, used in paints.
6.2 Stoichiometric Balance
To achieve high molecular weights, the stoichiometric ratio of functional groups must be close to 1:1. Even a small imbalance drastically reduces achievable chain length.
6.3 Catalysts and Reaction Conditions
Condensation reactions often require:
- Catalysts: acid catalysts (H₂SO₄, Ti(OBu)₄) or base catalysts (amines, metal oxides).
- High temperature: to remove by-products (e.g., water) and drive the equilibrium forward.
- Vacuum or inert gas flow: to assist in the removal of condensate and prevent oxidation.
7. Step-Growth vs Chain-Growth Polymerisation
| Feature | Step-Growth | Chain-Growth |
| Monomer types | Bi- or multifunctional monomers | Unsaturated monomers (C=C) |
| Growth mechanism | Stepwise reactions between any species | Growth from the active centre |
| By-products | Often (e.g. H₂O, HCl) | None |
| Molecular weight build-up | Gradual, requires high conversion | Rapid, early reaction |
| Examples | Polyesters, polyamides | Polyethene, polystyrene |
This distinction helps in predicting reaction control strategies and the required processing conditions.
8. Controlling Polymer Properties in Step-Growth Systems
8.1 Chain Length and Molecular Weight
Longer chains yield improved mechanical strength, higher melting points, and greater viscosity. Controlling reaction time and stoichiometry enables the tailoring of these features.
8.2 Crystallinity
Polyamides and polyesters can form semi-crystalline structures due to regular backbones and hydrogen bonding. Crystallinity affects transparency, barrier properties, and rigidity.
8.3 Copolymerisation and Modification
Copolymers can be formed by using mixed diacids or diols, modifying properties such as flexibility, melting point, or solubility. For instance, including adipic acid units in PET reduces its melting temperature, producing copolyester elastomers.
9. Case Studies in Step-Growth Polymers
9.1 Poly(ethylene terephthalate) (PET)
- Monomers: ethylene glycol + terephthalic acid
- Mechanism: esterification and polycondensation
- Properties: high strength, chemical resistance, transparency
- Applications: bottles, textiles (as polyester fibre), films
Industrial note: PET synthesis often involves an intermediate stage forming bis(hydroxyethyl) terephthalate (BHET), followed by melt polycondensation under vacuum.
9.2 Nylon-6,6
- Monomers: hexamethylene diamine + adipic acid
- Mechanism: amide condensation
- Properties: high tensile strength, abrasion resistance, high melting point
- Applications: engineering plastics, fibres, mechanical parts
Production: Conducted as a “nylon salt” solution polymerised under heat and pressure, yielding a tough, crystalline polymer.
9.3 Polycarbonates
- Monomers: bisphenol A + phosgene or diphenyl carbonate
- Mechanism: step-growth condensation producing carbonate linkages
- Properties: optical clarity, impact resistance
- Applications: lenses, electronics, safety equipment
10. Reaction Engineering and Process Techniques
10.1 Batch and Continuous Systems
- Batch reactors: suitable for small-scale or speciality polymers.
- Continuous reactors: employed for large-scale manufacture (e.g., PET, nylon).
10.2 Removal of Condensate
Efficient removal of by-products drives the equilibrium forward. This may involve:
- Vacuum distillation
- Inert gas stripping (e.g. nitrogen sweep)
- Azeotropic distillation
10.3 Melt and Solution Polymerisation
- Melt polymerisation: monomers are reacted without solvents; economical but requires high temperatures.
- Solution polymerisation: solvents assist heat transfer and by-product removal, but must be recovered later.
11. Mathematical Modelling of Step-Growth Polymerisation
11.1 Degree of Polymerisation (DP)
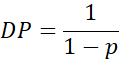
as derived by Carothers.
11.2 Number-Average Molecular Weight
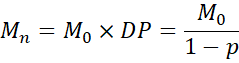
where Mo is the molecular weight of the repeating unit.
11.3 Polydispersity
Since polymer chains vary in length, step-growth polymers exhibit broad molecular weight distributions, typically with PDI ≈ 2 for ideal reactions.
12. Properties and Applications
Step-growth polymers exhibit a range of useful characteristics:
| Polymer | Major functional group | Key properties | Typical uses |
| Polyesters (PET) | –COO– | Transparent, strong, recyclable | Bottles, fibres |
| Polyamides (nylon) | –CONH– | Strong, abrasion-resistant | Textiles, gears |
| Polyurethanes | –NHCOO– | Flexible, elastic, durable | Foams, coatings |
| Epoxy resins | –C–O–C– (cross-linked) | Adhesive, chemically resistant | Composites, electronics |
| Alkyd resins | Ester network | Glossy, cross-linked | Paints, varnishes |
13. Step-Growth Polymers in Sustainability Context
13.1 Recycling
Certain step-growth polymers, such as PET and polyamides, can be chemically depolymerised back to their monomers (via hydrolysis or alcoholysis) and then repolymerised, forming the basis of chemical recycling.
13.2 Bio-Based Alternatives
Monomers like bio-derived ethylene glycol and succinic acid enable renewable versions of PET and polyesters. Biobased polyamides from castor oil (nylon-11, nylon-12) are gaining industrial traction.
13.3 Degradation and Biocompatibility
Aliphatic polyesters (e.g. poly(lactic acid), polycaprolactone) are biodegradable and biocompatible, making them suitable for medical sutures, implants, and controlled drug release systems.
14. Advantages and Limitations
Advantages:
- Broad selection of monomers and functional groups.
- Simple reaction apparatus.
- Controlled mechanical and chemical properties through monomer design.
- Potential for recyclability and bio-based sourcing.
Limitations:
- Requires near-complete reaction to achieve high molecular weight.
- Often slow compared with chain-growth systems.
- Removal of small-molecule by-products complicates processing.
- Thermal degradation is possible at high temperatures.
15. Experimental Example: Laboratory Synthesis of Nylon-6,6
A classic educational demonstration of step-growth polymerisation involves the nylon rope trick:
Procedure summary:
- Prepare an aqueous solution of hexamethylene diamine.
- Layer on top a solution of adipoyl chloride in an organic solvent (e.g. hexane).
- At the interface, an instantaneous reaction forms a thin nylon film.
- The polymer can be continuously pulled from the surface as a thread.
This visually striking experiment illustrates the essence of step-growth polymerisation at room temperature.
16. Analytical Characterisation
Characterisation confirms successful polymer formation and provides insight into structure:
| Technique | Observation | Application |
| Infrared spectroscopy (IR) | C=O stretching (~1730 cm⁻¹ in esters) | Identifies functional groups |
| NMR spectroscopy | Chemical shift of –OCH₂– or –NH– signals | Confirms monomer incorporation |
| Gel permeation chromatography (GPC) | Molecular weight distribution | Assesses chain length |
| Differential scanning calorimetry (DSC) | Tg and Tm values | Determines thermal behaviour |
| Thermogravimetric analysis (TGA) | Decomposition profile | Measures thermal stability |
17. Industrial Significance
Step-growth polymers underpin major industrial sectors:
- Textiles: nylon, polyester, spandex.
- Packaging: PET bottles and films.
- Automotive: polyamides for components and coatings.
- Electronics: epoxy resins and polyimides.
- Construction: polyurethane foams and sealants.
Production volumes exceed tens of millions of tonnes annually, highlighting their economic and practical importance.
18. Summary and Key Takeaways
- Step-growth polymerisation involves the stepwise reaction of multifunctional monomers, often producing small molecule by-products.
- Carothers’ equation links the extent of reaction to chain length, showing that high conversion is vital for polymer formation.
- Major step-growth polymers include polyesters, polyamides, polyurethanes, and epoxy resins.
- Reaction control, stoichiometry, and removal of condensate are essential for achieving desired molecular weights.
- Step-growth systems dominate key industries and increasingly support sustainable materials innovation.
This understanding provides the necessary foundation for Lecture 4, which will explore the complementary mechanism chain-growth polymerisation, particularly free-radical processes.
19. Further Reading and Learning Links
- Polymer Science Learning: Centre Condensation Polymerisation
- Royal Society of Chemistry: Step-Growth Mechanisms
- Khan Academy: Polymerisation Reactions
- Science History Institute: Wallace Carothers and Nylon
- Chemguide: Polyamides and Polyesters
These resources include diagrams, animations, and case studies that complement this lecture’s concepts.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections