Introduction
This lecture is designed to consolidate understanding of the quantitative chemistry concepts from Lectures 1-8. Students will apply:
- The mole concept and Avogadro’s number
- Molar mass calculations
- Mass-to-mole and mole-to-mass conversions
- Solution concentration calculations
- Percentage composition
- Empirical and molecular formula determination
- Balancing chemical equations
The aim is to link theory to practical problem-solving, building confidence for exams, laboratory work, and applied chemistry.
Example 1: Determining Moles from Mass
Problem: How many moles are in 50 g of aluminium sulphate, Al₂(SO₄)₃?
Step 1: Calculate molar mass:
- Al₂ = 2 × 26.98 = 53.96 g
- S₃ = 3 × 32.06 = 96.18 g
- O₁₂ = 12 × 16.00 = 192.00 g
- Total = 342.14 g/mol
Step 2: Use the formula:
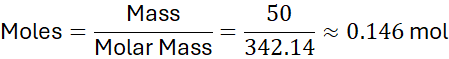
Step 3: Interpretation:
50 g of Al₂(SO₄)₃ contains approximately 0.146 moles of formula units.
Example 2: Calculating Mass from Moles
Problem: Calculate the mass of 0.75 moles of calcium hydroxide, Ca(OH)₂.
Step 1: Molar mass:
- Ca = 40.08 g
- O₂ = 2 × 16 = 32 g
- H₂ = 2 × 1.008 = 2.016 g
- Total = 74.096 g/mol
Step 2: Mass calculation:
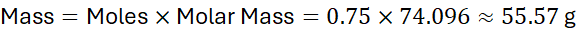
Answer: 55.57 g of Ca(OH)₂.
Example 3: Percentage Composition Application
Problem: A compound contains 45.5 g of P, 9.1 g of H, and 45.4 g of O. Determine the percentage composition.
Step 1: Total mass = 45.5 + 9.1 + 45.4 = 100 g
Step 2: Calculate percentages:
- P: 45.5%
- H: 9.1%
- O: 45.4%
Step 3: Interpretation:
Percentage composition confirms mass fractions of elements in the compound.
Example 4: Empirical Formula Determination
Problem: A compound contains 40% C, 6.7% H, and 53.3% O. Determine the empirical formula.
Step 1: Assume 100 g: C = 40 g, H = 6.7 g, O = 53.3 g
Step 2: Convert to moles:
- C: 40 / 12.01 ≈ 3.33
- H: 6.7 / 1.008 ≈ 6.65
- O: 53.3 / 16 ≈ 3.33
Step 3: Divide by the smallest:
- C = 1, H ≈ 2, O = 1
Step 4: Empirical formula = CH₂O
Example 5: Molecular Formula Determination
Problem: Compound has empirical formula CH₂O and molar mass 180 g/mol. Determine molecular formula.
Step 1: Empirical formula mass = 12 + 2 + 16 = 30 g/mol
Step 2: Calculate n: 180 / 30 = 6
Step 3: Multiply subscripts by n → C₆H₁₂O₆
Answer: Molecular formula = C₆H₁₂O₆
Example 6: Solution Concentration Calculation (Molarity)
Problem: 5.85 g of NaCl is dissolved in 250 mL of water. Determine molarity.
Step 1: Moles of NaCl:
Molar mass = 22.99 + 35.45 = 58.44 g/mol
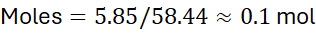
Step 2: Convert volume to litres: 0.250 L
Step 3: Molarity:
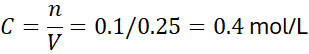
Answer: 0.4 M solution.
Example 7: Combining Mass and Percentage Composition
Problem: 10 g of MgSO₄·7H₂O is analysed. Find the mass of water.
Step 1: Molar mass of MgSO₄·7H₂O = 24.31 + 32.06 + 4 × 16 + 7 × 18.016 ≈ 246.47 g/mol
Water fraction = (7 × 18.016)/246.47 ≈ 0.511
Step 2: Mass of water = 0.511 × 10 g ≈ 5.11 g
Answer: 5.11 g of water.
Example 8: Reaction Stoichiometry
Problem: 25 g of H₂ reacts with excess O₂. How much H₂O is formed?
Step 1: Moles of H₂: 25 / 2.016 ≈ 12.4 mol
Reaction: 2 H₂ + O₂ → 2 H₂O → 1:1 mole ratio H₂:H₂O
Step 2: Moles H₂O = 12.4 mol
Step 3: Mass of H₂O = 12.4 × 18.016 ≈ 223.4 g
Answer: 223.4 g H₂O produced.
Example 9: Mass-to-Mass Calculation via Balanced Equation
Problem: How much Al₂O₃ is produced when 54 g of Al reacts with O₂?
Step 1: Reaction: 4 Al + 3 O₂ → 2 Al₂O₃
Step 2: Moles Al = 54 / 26.98 ≈ 2 mol
Step 3: Ratio Al : Al₂O₃ = 4:2 → 2 mol Al → 1 mol Al₂O₃
Step 4: Mass Al₂O₃ = 1 × (2 × 26.98 + 3 × 16) = 101.96 g
Answer: 101.96 g Al₂O₃
Example 10: Limiting Reactant
Problem: 10 g H₂ reacts with 80 g O₂. Determine the limiting reactant and mass of H₂O formed.
Step 1: Moles:
- H₂ = 10 / 2.016 ≈ 4.96 mol
- O₂ = 80 / 32 ≈ 2.5 mol
Step 2: Reaction: 2 H₂ + O₂ → 2 H₂O
- Required H₂ for 2.5 mol O₂ = 5 mol → only 4.96 mol H₂ available → H₂ is limiting
Step 3: Moles H₂O produced = 4.96 mol × (2/2) = 4.96 mol
Mass = 4.96 × 18.016 ≈ 89.4 g
Answer: 89.4 g H₂O, H₂ limiting.
Example 11: Dilution of Solutions
Problem: How to prepare 500 mL of 0.2 M NaOH from a 2 M stock solution?
Step 1: Use the dilution formula: C₁V₁ = C₂V₂
2 × V₁ = 0.2 × 0.5 → V₁ = 0.05 L = 50 mL
Step 2: Procedure: Mix 50 mL stock with water to 500 mL.
Example 12: Mass Percent Verification
Problem: Experimental sample of CuSO₄·5H₂O contains 24.6% Cu. Theoretical % Cu = 25.44%. Is the sample pure?
Step 1: Compare %: 24.6 < 25.44 → slightly impure, small loss of Cu or water.
Summary of Worked Examples
This lecture demonstrated:
- Converting mass ↔ moles
- Calculating percentage composition
- Determining empirical and molecular formulas
- Concentration and solution calculations
- Stoichiometric mass-to-mass calculations
- Limiting reactant and dilution problems
These examples integrate all quantitative chemistry concepts from previous lectures.
Next Lecture (Lecture 10): Worked Examples Part 2 – Advanced Multi-Step Problems Including Reactions in Series, Yields, and Industrial Applications.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections


