Introduction
Balancing chemical equations is a fundamental skill in chemistry. A balanced equation ensures that:
- The law of conservation of mass is obeyed
- The same number of atoms of each element appears on both sides
- Stoichiometric calculations for reactants and products are accurate
In this lecture, we cover:
- The principles of chemical equation balancing
- Step-by-step methods
- Worked examples of increasing complexity
- Applications in the laboratory and industry
- Common pitfalls and tips for accuracy
By the end of this lecture, students will be able to balance any chemical equation systematically and apply these skills in quantitative chemistry.
1. Principles of Balancing Equations
- Conservation of Mass: Atoms are neither created nor destroyed in a chemical reaction.
- Conservation of Charge: For reactions involving ions, the total charge must remain constant.
- Use of Coefficients: Only coefficients (numbers before compounds) can be adjusted to balance equations; subscripts (within chemical formulas) must not be changed.
2. Step-by-Step Method for Balancing
- Write the unbalanced equation using correct chemical formulas.
- List the number of atoms of each element on both sides.
- Balance one element at a time using coefficients.
- Balance hydrogen and oxygen last, as they often appear in multiple compounds.
- Check your work by counting atoms for each element.
- Simplify coefficients, if necessary, by dividing by the greatest common factor.
3. Worked Examples – Single Displacement Reactions
Example 1: Reaction of Zinc and Hydrochloric Acid
Unbalanced Equation:
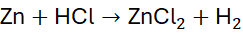
Step 1: List atoms:
- Zn: 1 → 1
- H: 1 → 2
- Cl: 1 → 2
Step 2: Balance H and Cl using a coefficient of 2 for HCl:
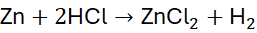
Step 3: Verify:
- Zn: 1 → 1
- H: 2 → 2
- Cl: 2 → 2
Balanced successfully.
Example 2: Reaction of Iron with Oxygen
Unbalanced Equation:
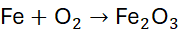
Step 1: Count atoms:
- Fe: 1 → 2
- O: 2 → 3
Step 2: Balance Fe first:
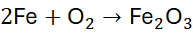
Step 3: Balance O by using the coefficient 3/2 for O₂:
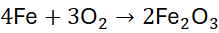
4. Worked Examples – Combustion Reactions
Example 3: Combustion of Propane (C₃H₈)
Unbalanced Equation:
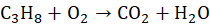
Step 2: Balance H: 4 H₂O
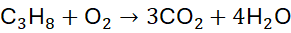
Step 3: Balance O: 3×2 + 4×1 = 10 → 5 O₂
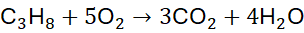
Example 4: Combustion of Butane (C₄H₁₀)
Unbalanced Equation:
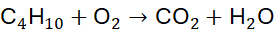
Step 1: Balance C: 4 CO₂
Step 2: Balance H: 5 H₂O
Step 3: Count O: 4×2 + 5×1 = 13 → 13/2 O₂
Step 4: Multiply the whole equation by 2:
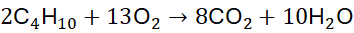
5. Worked Examples – Double Displacement Reactions
Example 5: Reaction of Silver Nitrate with Sodium Chloride
Unbalanced Equation:
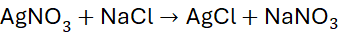
Step 1: List atoms: all elements appear once.
Step 2: Add coefficients if needed: 1:1:1:1
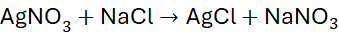
Already balanced.
Example 6: Reaction of Barium Chloride and Sulfuric Acid
Unbalanced Equation:
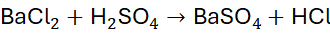
Step 1: Balance H and Cl: H₂Cl₂ → 2 HCl
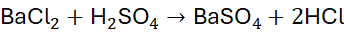
6. Worked Examples – Redox Reactions
Example 7: Reaction of Hydrogen Peroxide Decomposition
Unbalanced Equation:
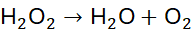
Step 1: Balance O: 2 H₂O + O₂?
Step 2: Balance H: H₂O requires 2 H₂O for 4 H → 2 H₂O + O₂
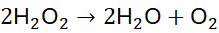
Example 8: Oxidation of Iron(II) to Iron(III)
Unbalanced Equation:
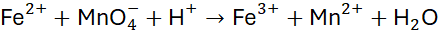
Step 1: Balance atoms and charges using the half-reaction method
Step 2: Overall balanced equation:
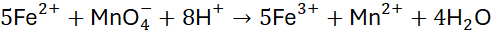
7. Common Errors and Pitfalls
- Changing subscripts instead of coefficients → alters the compound
- Ignoring diatomic elements (H₂, O₂, N₂)
- Balancing polyatomic ions separately instead of as units
- Forgetting to multiply through when fractional coefficients are used
8. Practical Tips for Balancing
- Start with elements that appear only once per side
- Balance polyatomic ions as a single unit
- Leave hydrogen and oxygen for last
- Check both the atom count and the charge if ionic species are involved
- Always verify by recounting all atoms
Further Reading:
9. Summary
Balancing chemical equations is an essential skill to ensure:
- Mass conservation
- Accurate stoichiometry
- Proper lab and industrial calculations
Key steps:
- Write the unbalanced equation
- Count the atoms of each element
- Adjust coefficients systematically
- Balance hydrogen and oxygen last
- Check and simplify
Balanced chemical equations provide the foundation for mole calculations, reaction yields, and quantitative analysis.
Next Lecture (Lecture 9): Worked Examples Part 1 – Applying Mass, Moles, Concentration, and Percentage Composition in Complex Problems.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections