From Natural Origins to Synthetic Mastery: The Story and Science of Macromolecules
1. Introduction: What Are Polymers?
Polymers are the molecular giants of chemistry. Derived from the Greek words poly (many) and meros (part), the term describes substances composed of many repeating structural units known as monomers. When monomers react chemically to form long, covalently bonded chains or networks, the resulting macromolecules display physical and chemical properties distinct from their individual components.
In daily life, polymers define much of the material world around us. The cellulose fibres in paper, the keratin in hair, the DNA that encodes genetic information, and the polyethene used in packaging are all polymers. Some occur naturally, others are engineered by human ingenuity, but all share a unifying principle: repetition gives rise to complexity.
Polymers are central to multiple scientific disciplines: chemistry, materials science, physics, engineering, biology, and environmental studies. Understanding their formation and behaviour provides the foundation for designing everything from biodegradable plastics to biomedical implants.
2. Historical Foundations: The Journey to Macromolecular Chemistry
2.1 Natural Polymers and Early Uses
Humanity’s relationship with polymers predates formal chemistry. Natural macromolecules such as silk, wool, and rubber were used for clothing, insulation, and waterproofing long before their molecular nature was understood. Ancient peoples exploited biopolymers such as cellulose (from plants) and collagen (from animal tissues) for textiles, glues, and early forms of paper and leather.
However, until the early twentieth century, chemists did not recognise these substances as composed of long, covalently bonded chains. They were often described as colloids or aggregates of small molecules, held together by ill-defined forces.
2.2 Staudinger and the Macromolecular Revolution
A turning point came in the 1920s when Hermann Staudinger proposed that rubber, cellulose, and other viscous organic materials were composed of giant molecules, macromolecules formed by the covalent linking of many monomer units. This idea met with scepticism; most chemists believed such long covalent chains would be unstable.
Through meticulous experimental work, Staudinger demonstrated that polymeric substances exhibited molecular weights far exceeding those of typical organic compounds yet behaved as single molecules in solution. His insight established the conceptual framework of polymer science and earned him the Nobel Prize in Chemistry in 1953.
2.3 The Synthetic Polymer Age
Once the existence of macromolecules was accepted, the search for synthetic polymers began. Several milestones followed:
- 1907 Bakelite: Leo Baekeland created phenol–formaldehyde resin, the first fully synthetic plastic.
- 1930s Nylon and Polyester: Wallace Carothers at DuPont synthesised nylon, a polymer that rivalled silk in strength and flexibility.
- 1940s–1950s Polyethene and Polypropylene: Karl Ziegler and Giulio Natta’s catalytic polymerisation methods enabled large-scale production of lightweight, durable plastics.
- 1970s onwards Advanced Functional Polymers: Conducting polymers, biodegradable plastics, and smart materials marked the diversification of polymer applications.
The discipline has since matured into one of the most interdisciplinary and industrially vital fields in modern science.
3. The Molecular Nature of Polymers
3.1 Chain Formation
A polymer chain consists of a sequence of repeating units, denoted as –[A]n–, where A represents the monomer unit and n the number of repetitions (degree of polymerisation). Depending on the monomer’s reactive sites, chains can be linear, branched, or cross-linked.
- Linear polymers (e.g. high-density polyethene) can pack efficiently, leading to crystalline regions.
- Branched polymers (e.g. low-density polyethene) pack less tightly, giving flexibility and lower melting points.
- Cross-linked polymers (e.g. vulcanised rubber, epoxy resins) form networks with high elasticity or rigidity, depending on cross-link density.
3.2 Molecular Weight and Polydispersity
Unlike small molecules, polymers have a distribution of chain lengths rather than a single molecular weight. This is expressed using two averages:
- Number-average molecular weight (Mn): the arithmetic mean of molecular weights.
- Weight-average molecular weight (Mw): gives greater weight to larger molecules.
Their ratio, Mw/Mn, defines the polydispersity index (PDI). A PDI of 1 indicates uniformity (rare in practice); most polymers range between 1.2 and 3.0.
Molecular weight affects strength, toughness, viscosity, and melting behaviour, hence its control is central to polymer synthesis.
4. Bonding and Structure
4.1 Covalent Framework
The backbone of a polymer is held together by covalent bonds, typically carbon–carbon or carbon–heteroatom linkages (C–O, C–N, C–S). The type of bond influences both chemical stability and flexibility.
4.2 Intermolecular Forces
While covalent bonds form the molecular skeleton, intermolecular forces define bulk properties:
- Van der Waals forces: dominate in non-polar polymers such as polyethene.
- Dipole–dipole interactions: significant in polar polymers like polyvinyl chloride (PVC).
- Hydrogen bonding: strengthens polymers containing –OH or –NH groups, as in nylon or cellulose.
The balance of these interactions determines melting point, tensile strength, and solubility.
4.3 Crystalline and Amorphous Domains
Most solid polymers contain both crystalline and amorphous regions. Crystallinity enhances mechanical strength and chemical resistance; amorphous regions add flexibility and transparency. The proportion depends on chain regularity, tacticity, and processing conditions. For example, polyethene terephthalate (PET) can form either clear (amorphous) or opaque (crystalline) bottles depending on how it is cooled.
5. Mechanisms of Polymer Formation
5.1 Step-Growth Polymerisation
In step-growth polymerisation, monomers with two or more reactive functional groups (e.g. –OH, –COOH, –NH₂) react in successive steps. Any two species, monomer, dimer, or trimer, can combine, releasing small molecules such as water or methanol.
Examples include:
- Polyesters (from diols and dicarboxylic acids)
- Polyamides (from diamines and dicarboxylic acids)
High molecular weight polymers form only when the reaction approaches completion, making stoichiometric balance crucial.
5.2 Chain-Growth Polymerisation
Chain-growth polymerisation proceeds via active centres (radicals, cations, anions, or metal complexes) that add monomers sequentially to a growing chain.
Three main steps occur:
- Initiation: generation of reactive species (e.g. free radicals).
- Propagation: successive addition of monomers to the active site.
- Termination: deactivation by combination or disproportionation.
Polystyrene, polyethene, and PVC are classic examples. The process allows rapid molecular weight build-up early in the reaction.
5.3 Copolymerisation
When two or more monomer species polymerise together, the result is a copolymer. Arrangement of the different monomer units can be:
- Random (statistical) units distributed irregularly
- Alternating strict alternation of A and B
- Block long sequences of one monomer followed by another
- Graft side chains of one polymer type attached to another backbone
Copolymers enable fine-tuning of flexibility, hardness, chemical resistance, and transparency.
6. Structural Parameters and Their Influence
6.1 Degree of Polymerisation (DP)
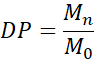
where Mn is the number-average molecular weight, and Mo is the monomer molecular weight. DP typically ranges from a few hundred to several thousand, depending on polymer type and synthesis method.
6.2 Tacticity
For polymers with asymmetric carbon centres (e.g. polypropylene), tacticity, the arrangement of substituents along the chain, determines crystallinity:
- Isotactic: substituents on the same side → crystalline, rigid.
- Syndiotactic: alternating arrangement → ordered, flexible.
- Atactic: random arrangement → amorphous, soft.
Tacticity is controlled by catalysts such as Ziegler–Natta or metallocenes.
6.3 Cross-Link Density
The extent of cross-linking affects elasticity and thermal resistance.
- Low cross-link density: soft elastomers.
- High cross-link density: rigid thermosets.
Vulcanised rubber exemplifies this balance, as sulphur atoms form bridges between chains, imparting resilience.
7. Physical Properties of Polymers
7.1 Thermal Behaviour
Two key transitions govern polymer thermal properties:
- Glass transition temperature (Tg): below this, the polymer is glassy and brittle; above it, rubbery and flexible.
- Melting temperature (Tm): the point at which crystalline regions melt, enabling flow.
Amorphous polymers (e.g. polystyrene) have only Tg; semi-crystalline ones (e.g. polyethene) show both Tg and Tm.
7.2 Mechanical Characteristics
Polymers exhibit diverse mechanical behaviours:
- Plastics: rigid or semi-rigid solids mouldable with heat (e.g. PVC, polycarbonate).
- Elastomers: highly extensible materials that return to shape (e.g. natural rubber, silicone).
- Fibres: long, oriented chains providing tensile strength (e.g. nylon, Kevlar).
Chain entanglement, crystallinity, and molecular weight determine these characteristics.
7.3 Solubility and Chemical Resistance
A polymer dissolves when the interaction between the polymer and solvent overcomes internal cohesive forces. Polar polymers dissolve in polar solvents; non-polar ones require non-polar solvents.
Cross-linked polymers are usually insoluble but may swell when solvents penetrate between chains.
8. Characterisation Techniques
Characterisation bridges the gap between chemical structure and macroscopic behaviour.
| Technique | Property Determined | Example Insight |
| Gel Permeation Chromatography (GPC) | Molecular weight distribution | Confirms polymer chain size uniformity |
| NMR & IR Spectroscopy | Functional groups and sequence | Identifies monomer composition |
| Differential Scanning Calorimetry (DSC) | Tg and Tm | Measures thermal transitions |
| X-Ray Diffraction (XRD) | Crystallinity | Quantifies ordered regions |
| Rheometry | Flow and deformation | Assesses viscosity and elasticity |
Together, these tools provide a detailed fingerprint of polymer architecture.
9. Classification Systems in Polymer Science
Chemists categorise polymers in several complementary ways:
9.1 By Origin
- Natural: cellulose, proteins, DNA, rubber.
- Synthetic: polyethene, nylon, acrylics.
- Semi-synthetic: cellulose acetate, rayon.
9.2 By Thermal Response
- Thermoplastics: soften on heating, remoldable (e.g. polypropylene).
- Thermosets: harden irreversibly on curing (e.g. epoxy resins).
- Elastomers: stretch and recover (e.g. silicone rubber).
9.3 By Structure
- Linear, branched, cross-linked, or network polymers.
9.4 By Composition
- Homopolymers (single monomer).
- Copolymers (two or more monomers).
9.5 By Polymerisation Mechanism
- Addition (chain-growth) versus condensation (step-growth).
10. Environmental Context and Sustainable Perspectives
10.1 The Dual Nature of Polymers
Polymers revolutionised modern life, but their durability has created environmental challenges. Conventional plastics persist for centuries, fragmenting into microplastics that contaminate ecosystems. Sustainable polymer chemistry seeks solutions through recycling, biodegradation, and renewable sourcing.
10.2 Biopolymers and Bio-Based Alternatives
Polymers like polylactic acid (PLA) and polyhydroxyalkanoates (PHAs), derived from corn starch or microbial fermentation, offer biodegradable alternatives. Advances in green catalysis and solvent-free processes are improving their viability.
10.3 Circular Economy and Chemical Recycling
Rather than discarding, future materials cycles aim to recover monomers for reuse via chemical recycling depolymerisation, followed by repolymerisation. This approach preserves molecular quality while reducing waste.
11. Quantitative Frameworks
Three equations provide conceptual scaffolding in polymer physical chemistry:
- Degree of Polymerisation (DP)
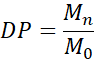
- Mark–Houwink Equation
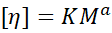
Relates intrinsic viscosity to molecular weight.
- Flory–Huggins Equation
Models polymer–solvent mixing behaviour through the interaction parameter (χ), predicting solubility and phase separation.
These relations connect experimental observables with theoretical insight.
12. Polymer Science as an Interdisciplinary Field
Polymer chemistry intersects with multiple domains:
- Biology: proteins, DNA, polysaccharides.
- Physics: rheology, crystallisation, and viscoelasticity.
- Engineering: materials design, composites, coatings.
- Medicine: hydrogels, sutures, and drug delivery systems.
- Electronics: conductive polymers and flexible circuits.
This interdisciplinarity fosters innovation across science and industry.
13. Current Research Frontiers
- Self-healing polymers: repairing microcracks autonomously.
- Stimuli-responsive materials: reacting to pH, light, or temperature.
- Reprocessable thermosets: combining durability with recyclability.
- Polymer nanocomposites: integrating nanoscale fillers for strength and conductivity.
- Computational polymer design: predicting structure–property relationships with artificial intelligence.
Such innovations demonstrate how polymer science is evolving towards functionality, adaptability, and sustainability.
14. Summary and Key Takeaways
- Polymers are macromolecules formed by repeating monomeric units.
- Their structure, bonding, and molecular weight govern physical properties.
- Polymerisation mechanisms (step-growth and chain-growth) determine chain architecture.
- Intermolecular forces, tacticity, and crystallinity shape performance.
- Polymers’ widespread use necessitates environmental responsibility and sustainable design.
Understanding these principles is essential before exploring more specific topics such as classification, synthesis, and characterisation, which will follow in subsequent lectures.
15. Further Reading and Learning Links
For visual explanations, molecular models, and historical context, explore the following reputable resources:
- Polymer Science: Learning Centre Basics
- Royal Society of Chemistry: Polymers
- Khan Academy: Macromolecules
- Science History Institute: Plastics and Polymers
- Chemguide: Polymerisation
Each provides interactive diagrams, animations, or accessible text that complements this lecture’s content.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections


