Introduction
Empirical and molecular formulas are essential in chemistry for describing the composition of compounds.
- The empirical formula shows the simplest whole-number ratio of atoms in a compound.
- The molecular formula shows the actual number of atoms of each element in a molecule.
Understanding these formulas is critical for:
- Identifying unknown compounds
- Confirming chemical synthesis results
- Performing stoichiometric calculations
- Analysing chemical composition in industry and research
This lecture covers:
- Definitions and differences between empirical and molecular formulas
- Step-by-step methods for calculation
- Worked examples of increasing complexity
- Applications in the laboratory, industry, and research
- Common pitfalls and tips for accuracy
By the end of this lecture, students will be able to determine both empirical and molecular formulas from experimental data and theoretical compositions.
1. Empirical Formula
1.1 Definition
The empirical formula represents the simplest ratio of moles of each element in a compound. It does not necessarily reflect the actual number of atoms in a molecule but provides a fundamental ratio.
1.2 Steps to Determine Empirical Formula
- Obtain mass or percentage composition of each element.
- Convert mass to moles using the atomic masses.
- Divide all mole values by the smallest number of moles to get a simple ratio.
- Multiply by a factor if necessary to achieve whole numbers.
2. Worked Examples – Empirical Formula
Example 1: Simple Compound
Problem: A compound contains 40% C, 6.7% H, and 53.3% O. Determine the empirical formula.
Solution:
- Assume 100 g of compound → C = 40 g, H = 6.7 g, O = 53.3 g
- Convert to moles:
- C: 40 / 12.01 ≈ 3.33 mol
- H: 6.7 / 1.008 ≈ 6.65 mol
- O: 53.3 / 16.00 ≈ 3.33 mol
- Divide by the smallest:
- C: 3.33 / 3.33 = 1
- H: 6.65 / 3.33 ≈ 2
- O: 3.33 / 3.33 = 1
- Empirical formula = CH₂O
Example 2: More Complex Compound
Problem: A compound contains 52.14% C, 34.73% O, and 13.13% H. Find the empirical formula.
Solution:
- Assume 100 g: C = 52.14 g, O = 34.73 g, H = 13.13 g
- Convert to moles:
- C: 52.14 / 12.01 ≈ 4.34 mol
- O: 34.73 / 16.00 ≈ 2.17 mol
- H: 13.13 / 1.008 ≈ 13.03 mol
- Divide by the smallest (2.17):
- C: 4.34 / 2.17 ≈ 2
- O: 2.17 / 2.17 = 1
- H: 13.03 / 2.17 ≈ 6
- Empirical formula = C₂H₆O
3. Molecular Formula
3.1 Definition
The molecular formula represents the actual number of atoms of each element in a molecule. The molecular formula is a whole-number multiple of the empirical formula:
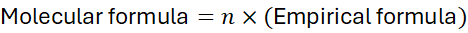
Where:
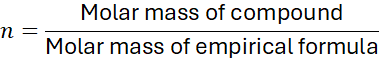
3.2 Steps to Determine Molecular Formula
- Determine the empirical formula.
- Calculate the molar mass of the empirical formula.
- Divide the molar mass of the compound by the empirical formula mass to find
.
- Multiply the subscripts in the empirical formula by
to obtain the molecular formula.
4. Worked Examples – Molecular Formula
Example 3: Simple Molecule
Problem: A compound has an empirical formula CH₂O and a molar mass of 180 g/mol. Find the molecular formula.
Solution:
- Empirical formula mass = 12 + 2 × 1 + 16 = 30 g/mol
- n = 180 / 30 = 6
- Molecular formula = C₆H₁₂O₆
Example 4: More Complex Molecule
Problem: Empirical formula = C₂H₄O, molar mass = 88 g/mol. Find molecular formula.
- Empirical formula mass = 2 × 12 + 4 × 1 + 16 = 44 g/mol
- n = 88 / 44 = 2
- Molecular formula = C₄H₈O₂
Example 5: Nitrogen-Containing Compound
Problem: A compound contains 38.7% C, 9.7% H, and 51.6% N. Molar mass = 80 g/mol. Find empirical and molecular formulas.
Solution:
- Assume 100 g: C = 38.7 g, H = 9.7 g, N = 51.6 g
- Convert to moles:
- C: 38.7 / 12.01 ≈ 3.22
- H: 9.7 / 1.008 ≈ 9.63
- N: 51.6 / 14.01 ≈ 3.68
- Divide by the smallest (3.22):
- C ≈ 1
- H ≈ 3
- N ≈ 1.14 → round to nearest whole number ratio = 1:3:1
- Empirical formula ≈ CH₃N
- Empirical formula mass = 12 + 3 + 14 = 29 g/mol
- n = 80 / 29 ≈ 2.76 → round to 3
- Molecular formula = C₃H₉N₃
5. Applications of Empirical and Molecular Formulas
- Determining Unknown Compounds: Combine experimental mass data with percentage composition to identify compounds.
- Confirming Synthesis: Verify that laboratory-produced compounds match expected formulas.
- Stoichiometry: Essential for balancing chemical equations and calculating reactant/product quantities.
- Pharmaceuticals: Ensuring the correct molecular formula ensures proper drug efficacy.
- Industrial Chemistry: Optimises production of polymers, fertilisers, and other chemicals.
6. Common Errors and Pitfalls
- Rounding too early: Maintain precision through all calculations.
- Ignoring significant figures: Follow standard rules of chemistry.
- Incorrect empirical formula: Carefully check mole ratios.
- Fractional subscripts: Multiply all subscripts to achieve whole numbers.
- Incorrect molar mass: Use accurate atomic masses.
7. Practical Tips
- Always start with percentage composition or mass data.
- Convert to moles before determining ratios.
- Divide by the smallest mole value for the simplest ratio.
- Verify molecular formula by comparing empirical formula mass to molecular mass.
- For fractional ratios, multiply by the lowest factor to obtain whole numbers.
Further Reading:
8. Summary of Steps
- Determine the percentage composition or mass of elements.
- Convert mass to moles.
- Determine the simplest ratio → empirical formula.
- Calculate empirical formula mass.
- Divide molar mass by empirical formula mass →
.
- Multiply empirical formula subscripts by
→ molecular formula.
9. Worked Examples Summary Table
| Example | Data | Empirical Formula | Molecular Formula |
| 1 | 40% C, 6.7% H, 53.3% O | CH₂O | C₆H₁₂O₆ |
| 2 | 52.14% C, 34.73% O, 13.13% H | C₂H₆O | C₂H₆O |
| 3 | CH₂O, molar mass 180 g/mol | CH₂O | C₆H₁₂O₆ |
| 4 | C₂H₄O, molar mass 88 g/mol | C₂H₄O | C₄H₈O₂ |
| 5 | 38.7% C, 9.7% H, 51.6% N, molar mass 80 g/mol | CH₃N | C₃H₉N₃ |
Conclusion
Empirical and molecular formulas provide essential insights into chemical composition:
- The empirical formula offers the simplest ratio of atoms.
- The molecular formula reflects the actual molecular structure.
Mastery of these calculations allows chemists to:
- Identify unknown compounds
- Verify chemical synthesis
- Perform stoichiometric calculations
- Support industrial and laboratory applications
Next Lecture (Lecture 8): Balancing Chemical Equations – Mass and Mole Relationships in Reactions.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections


