Introduction
Percentage composition is a fundamental concept in quantitative chemistry. It allows chemists to determine the proportion of each element in a compound by mass. Understanding percentage composition is essential for:
- Calculating empirical and molecular formulas
- Verifying the purity of compounds
- Comparing theoretical and experimental data
- Preparing solutions and reagents accurately
This lecture covers:
- Definition of percentage composition
- Step-by-step calculation method
- Worked examples with increasing complexity
- Applications in the laboratory and industry
- Common errors and tips for accuracy
By the end of this lecture, students will be able to calculate and interpret the mass percentage of each element in any chemical compound.
1. Definition of Percentage Composition
The percentage composition of an element in a compound expresses the mass of the element as a percentage of the total mass of the compound.
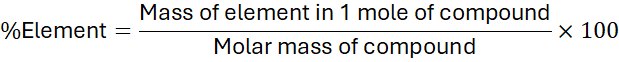
Where:
- Mass of element = number of atoms × atomic mass
- Molar mass = sum of all atomic masses in the compound
2. Step-by-Step Calculation Method
Step 1: Determine the molar mass of the compound
Use the periodic table to sum the atomic masses of all atoms in the compound.
Step 2: Calculate the mass of each element in one mole
Multiply the number of atoms of each element by its atomic mass.
Step 3: Divide by the molar mass
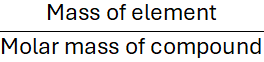
Step 4: Multiply by 100 to get the percentage
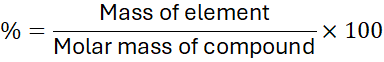
3. Worked Examples with Increasing Complexity
Example 1: Water (H₂O)
Problem: Calculate the percentage composition of H₂O.
- Molar mass of H₂O = 2 × 1.01 + 16.00 = 18.02 g/mol
- Hydrogen = 2 × 1.01 = 2.02 g
- Oxygen = 16.00 g
Percentage composition:
- Hydrogen: (2.02 ÷ 18.02) × 100 ≈ 11.21%
- Oxygen: (16.00 ÷ 18.02) × 100 ≈ 88.79%
Example 2: Carbon Dioxide (CO₂)
- Molar mass of CO₂ = 12.01 + 32.00 = 44.01 g/mol
- Carbon = 12.01 g
- Oxygen = 32.00 g
Percentage composition:
Oxygen: (32.00 ÷ 44.01) × 100 ≈ 72.71%
Carbon: (12.01 ÷ 44.01) × 100 ≈ 27.29%
Example 3: Glucose (C₆H₁₂O₆)
- Molar mass = 6 × 12.01 + 12 × 1.008 + 6 × 16.00 = 180.16 g/mol
- Carbon: 72.06 g
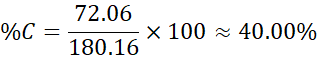
- Hydrogen: 12.096 g
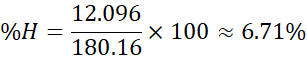
- Oxygen: 96.00 g
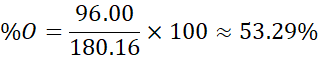
Example 4: Sulfuric Acid (H₂SO₄)
- Molar mass = 2 × 1.008 + 32.06 + 4 × 16.00 = 98.08 g/mol
- Hydrogen: 2.016 g → 2.06%
- Sulphur: 32.06 g → 32.69%
- Oxygen: 64.00 g → 65.25%
Example 5: Ammonium Nitrate (NH₄NO₃)
- Molar mass = 14.01 + 4 × 1.008 + 14.01 + 3 × 16.00 = 80.04 g/mol
- Nitrogen: 28.02 g
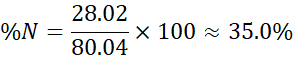
- Hydrogen: 4.032 g → 5.04%
- Oxygen: 48.00 g → 59.96%
Example 6: Magnesium Hydroxide Mg(OH)₂
- Molar mass = 24.31 + 2 × (16 + 1.008) = 58.33 g/mol
- Mg: 24.31 g → 41.7%
- O: 32.00 g → 54.9%
- H: 2.016 g → 3.5%
Example 7: Calcium Carbonate (CaCO₃)
- Molar mass = 40.08 + 12.01 + 3 × 16.00 = 100.09 g/mol
- Ca: 40.08 g → 40.05%
- C: 12.01 g → 12.0%
- O: 48.00 g → 47.95%
Example 8: Copper(II) Sulphate Pentahydrate (CuSO₄·5H₂O)
- Molar mass = 63.55 + 32.06 + 4 × 16.00 + 5 × 18.016 = 249.7 g/mol
- Cu: 63.55 g → 25.44%
- S: 32.06 g → 12.85%
- O: 64 + 90.08 = 154.08 g → 61.71%
Example 9: Iron(III) Oxide (Fe₂O₃)
- Molar mass = 2 × 55.85 + 3 × 16.00 = 159.7 g/mol
- Fe: 111.7 g → 69.97%
- O: 48 g → 30.03%
Example 10: Sodium Sulphate (Na₂SO₄)
- Molar mass = 2 × 22.99 + 32.06 + 4 × 16.00 = 142.04 g/mol
- Na: 45.98 g → 32.4%
- S: 32.06 g → 22.6%
- O: 64 g → 45.0%
Example 11: Potassium Permanganate (KMnO₄)
- Molar mass = 39.10 + 54.94 + 4 × 16 = 158.04 g/mol
- K: 39.10 g → 24.76%
- Mn: 54.94 g → 34.78%
- O: 64 g → 40.46%
Example 12: Ethanol (C₂H₅OH)
- Molar mass = 2 × 12.01 + 6 × 1.008 + 16.00 = 46.08 g/mol
- C: 24.02 g → 52.15%
- H: 6.048 g → 13.12%
- O: 16 g → 34.73%
4. Applications of Percentage Composition
- Empirical and Molecular Formula Determination: Use mass percentages to calculate mole ratios.
- Purity Verification: Compare experimental vs theoretical percentages.
- Chemical Synthesis: Ensure reactants are mixed in correct ratios.
- Quality Control: Monitor industrial processes, pharmaceuticals, and alloys.
- Environmental Chemistry: Determine pollutant composition in samples.
5. Common Errors
- Forgetting to multiply by the number of atoms per formula unit
- Using incorrect atomic masses
- Rounding too early, causing cumulative errors
- Ignoring hydrates or water of crystallisation
6. Practical Tips
- Always calculate the molar mass first before percentages
- Check formula unit counts carefully
- Keep a stepwise method: mass per element → divide by total → multiply by 100
- Use a periodic table with decimals for precise results
Further Reading:
Conclusion
Percentage composition is a versatile tool in quantitative chemistry. Mastery of this concept enables students to:
- Determine the proportion of elements in compounds
- Verify chemical purity and composition
- Link experimental data to theoretical calculations
- Support further calculations, including empirical and molecular formulas
Next Lecture (Lecture 7): Empirical and Molecular Formulas – Determining Simplest and Actual Ratios of Atoms.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections


