Introduction
In chemistry, solutions are ubiquitous, from laboratory experiments to industrial processes. Understanding the concentration of a solution is essential for controlling reactions, ensuring reproducibility, and achieving desired chemical effects. Concentration links the amount of solute to the volume of solvent, often using the mole as a standard unit.
This lecture covers:
- Definition of solution concentration
- Molarity and its calculation
- Dilution principles and calculations
- Step-by-step worked examples
- Common units of concentration
- Practical applications in the laboratory and industry
- Common errors and tips
By the end of this lecture, students will be able to calculate concentrations, perform dilutions, and apply concentration concepts to a wide range of chemical problems.
1. Definition of Solution Concentration
Concentration is a measure of how much solute is present in a given quantity of solvent or solution.
- Expressed as moles per litre (molarity, M) or as mass per volume (g/L, mg/mL).
- Central to stoichiometry, as it links the amount of substance to the volume of solution.
Basic formula for molarity:
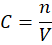
Where:
= concentration (mol/L)
= number of moles of solute (mol)
= volume of solution (L)
2. Molarity
Molarity (M) is the number of moles of solute per litre of solution. It is the most common unit of concentration in chemistry.
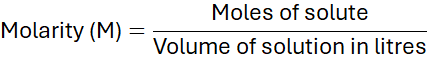
2.1 Calculating Molarity
Example 1:
Calculate the molarity of a solution containing 0.5 moles of NaCl in 0.25 L of water.
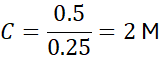
2.2 Calculating Moles from Molarity
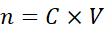
Example 2:
How many moles are in 0.75 L of 0.2 M H₂SO₄?
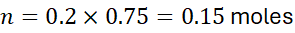
2.3 Calculating Mass from Molarity
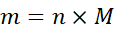
Example 3:
Find the mass of NaOH required to make 500 mL of 1 M solution.
Solution:
- Volume of solution = 500 mL = 0.5 L
- Molarity (M) = 1 M
Moles of NaOH = Molarity × Volume = 1 × 0.5 = 0.5 moles
Molar mass of NaOH = 23 + 16 + 1 = 40 g/mol
Mass of NaOH = moles × molar mass = 0.5 × 40 = 20 g
3. Dilution of Solutions
Dilution reduces the concentration of a solution by adding more solvent. The total moles of solute remain unchanged.
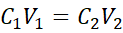
Where:
- C₁ = initial concentration
- V₁ = initial volume
- C₂ = final concentration
- V₂ = final volume
Example 4:
Dilute 100 mL of 2 M HCl to 500 mL. Find the new concentration.
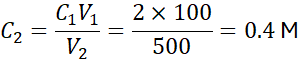
3.1 Serial Dilutions
Serial dilutions are performed when very low concentrations are required. Each step reduces the concentration by a known factor.
Example 5:
Diluting a 1 M solution by a factor of 10 three times:
- Step 1: 1 M → 0.1 M
- Step 2: 0.1 M → 0.01 M
- Step 3: 0.01 M → 0.001 M
4. Mass per Volume Concentrations
Sometimes concentration is expressed in g/L, especially in industrial applications.
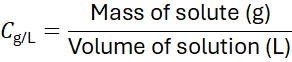
Example 6:
A solution contains 10 g of KCl in 250 mL. Calculate concentration in g/L.
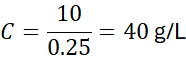
5. Preparing Solutions
5.1 Steps for Laboratory Preparation
- Calculate the moles or mass of solute required.
- Measure the solute accurately using a balance.
- Add solute to a volumetric flask.
- Add solvent gradually, mixing thoroughly.
- Make up to the mark for precise volume.
Example 7:
Prepare 250 mL of 0.5 M Na₂SO₄:
- Molar mass Na₂SO₄ = 142 g/mol
- Moles required = 0.5 × 0.25 = 0.125 mol
- Mass = 0.125 × 142 ≈ 17.75 g
- Dissolve in water and make up to 250 mL
5.2 Tips for Accuracy
- Use a volumetric flask for precise measurements.
- Stir or invert the solution to ensure uniformity.
- Account for temperature effects on volume if necessary.
6. Worked Examples with Increasing Complexity
Example 8: Concentration after Dilution
Problem: 50 mL of 3 M H₂SO₄ is diluted to 250 mL. What is the new concentration?
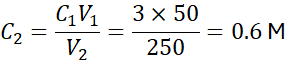
Example 9: Number of Moles in a Volume of Solution
Problem: How many moles are in 100 mL of 0.1 M NaOH?
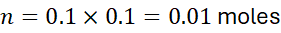
Example 10: Mass of Solute for Specific Molarity
Problem: Prepare 200 mL of 0.2 M CaCl₂.
- Moles = 0.2 × 0.2 = 0.04 mol
- Molar mass = 40.08 + 2 × 35.45 = 110.98 g/mol
- Mass = 0.04 × 110.98 ≈ 4.44 g
Example 11: Mass per Volume Calculation
Problem: A 0.25 M Na₂CO₃ solution contains 26.5 g of Na₂CO₃. What volume is this solution?
- Moles = 26.5 / 105.99 ≈ 0.25 mol
- Volume = moles / concentration = 0.25 / 0.25 = 1 L
Example 12: Serial Dilution to Achieve Low Concentration
Problem: Prepare 0.01 M HCl from 1 M stock solution.
- Required dilution factor = 1 / 0.01 = 100
- Take 10 mL stock and dilute to 1 L
7. Common Units of Concentration
- Molarity (M) = mol/L
- Mass/volume (g/L, mg/mL)
- Percentage solutions (% w/v, % v/v)
- Molality (mol/kg solvent) – less common but useful for temperature-dependent studies
Further Reading:
8. Common Errors and How to Avoid Them
- Unit confusion: Always convert mL to L for molarity calculations.
- Ignoring dilution factors: Moles of solute do not change during dilution.
- Measurement inaccuracies: Use proper laboratory glassware.
- Rounding errors: Maintain significant figures throughout calculations.
9. Practical Applications
- Laboratory: Preparing buffers, titrations, and reaction solutions
- Industry: Producing chemical products with precise concentrations
- Pharmaceuticals: Accurate dosing in medicinal solutions
- Environmental chemistry: Determining pollutant concentrations in water
Conclusion
Understanding solution concentration is vital for both theoretical and practical chemistry. By mastering:
- Molarity calculations
- Dilution principles
- Mass per volume and percentage concentrations
…students can perform accurate laboratory work, solve stoichiometric problems, and apply these concepts in industrial and environmental contexts.
Next Lecture (Lecture 6): Percentage Composition – Determining Elemental Mass Percentages in Compounds.
Support the Archive
This archive is freely shared as a communal act of care.
If you’d like to support its continuation, consider purchasing a companion PDF set for £1 per lecture, with an associated quiz, via Payhip, with the final price depending on the number of lectures in the set, available only once the full series is complete.
Explore more with us:
- Read our Informal Blog for relaxed insights
- Discover Deconvolution and see what’s happening
- Visit Gwenin for a curated selection of frameworks
- Browse Spiralmore collections


